| Plants |
| Theory |
| Programs & Download |
| Ideas Exchange |
| Sponsors | |
|
|
TOWER
- Calculate of the haulage of liquids for the vapor
- Calculate of the flooding of the trays
PIPES
- Calculate of the decibels taken place by the step of the fluid in the pipe
PUMPS
- Calculate of the minimum pressure before the cavitation
VALVES
- Calculate of the minimum pressure before the cavitation
FURNACES
- Calculate the consumption of the fuel
- Calculate of the reactions happened among of the tubes
BOILERS
- Calculate of the sure diameter of the pipe of the boiler
- Calculate of the lost of heat of the boiler
- Calculate of the reactions happened by the contamination
HEAT EXCHANGERS
- Calculate of the temperature of the tube
- Calculate of the flow with phase change.
Density - program online for pure compound
For liquids
to obtain the density of a liquid the following formula is used:
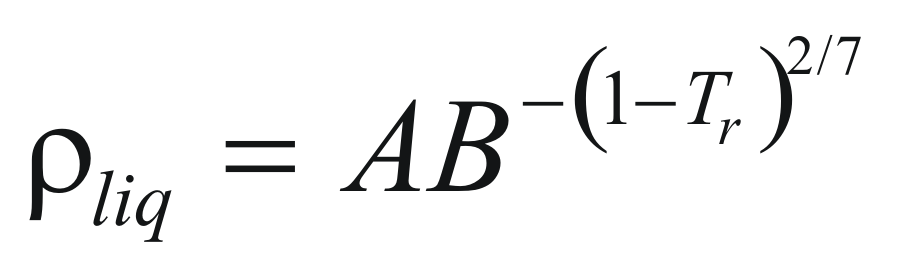
Where ρliq is the density of the pure liquid (gr/cm3)
A and B are constant that they depend on the compound.
Tr is the reduced temperature.
For the Gases
to obtain the density of a gas the following formula is used:
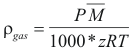
Where ρgas is the density of the gas (gr/cm3)
P is the total pressure (Atm)
M is the molecular weight (gr).
z is the factor of compressibility of the gas.
T is the temperature of the gas (°K)
R is the constant of the gas = 0.082
For the Mixture
to obtain the density average of the mixture the following formula is used
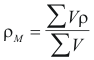
Where ρM is the density average of the mixture (gr/cm3)
ρ is the density of the component i (gr/cm3)
V is the volume that occupies according to its fraction molar (cm3)
Special case in density of hydrocarbons
Viscosity - program online for pure compound
; For the liquids
to obtain the viscosity of a liquid the following formula is used:
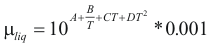
Where μliq is the viscosity of the liquid (kg/m*s)
A, B, C and D are constant that it depends on the compound
T is the temperature (°K)
For the gases
to obtain the viscosity of a gas the following formula is used:
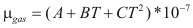
Where μgas is the viscosity of the gas (kg/m*s)
A, B and C are constant that it depends on the compound
T is the temperature (°K)
For the Mixture
to obtain the viscosity of the mixture the following formula is used
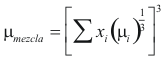
Where μmixture is the viscosity of the mixture (kg/m*s)
μi is the viscosity of each compound (kg/m*s)
xi is the fraction molar of the mixture
Caloric capacity
For the liquids
to obtain the caloric capacity of a liquid the following formula is used:
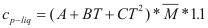
Where Cp-liq is the caloric capacity of a pure liquid (cal/mol-°K).
A, B and C are constant that they depend on the compound
T is the temperature to which is the liquid. (°K)
M is the molecular weight of the compound (gr).
For the gases
to obtain the caloric capacity of a gas the following formula is used:
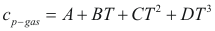
Where Cp-gas is the caloric capacity of a gas (cal/mol-°K).
A, B, C and D are constant that they depend on the compound
T is the temperature. (°K)
For the mixture
to obtain the caloric capacity the mixture the following formula is used:
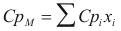 o
o
Where CpM is the caloric capacity of the mixture (cal/mol°K)
Cpi is the caloric capacity of the component i (cal/mol°K).
xi is the fraction molar of the component i
Thermal Conductivity
For the liquids
to obtain the thermal conductivity of a liquid the following formula is used:
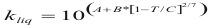
Where kliq is the thermal conductivity of the pure liquid (w/m°K)
A, B, C is constant that they depend on the liquid
T is the temperature (°K)
For the gases
to obtain the thermal conductivity of a gas the following formula is used:
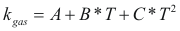
Where kgas is the thermal conductivity of the gas (w/m°K)
A, B, C is constant that it depends on the gas.
T is the temperature (°K)
For the mixture
to obtain the thermal conductivity of the mixture the following formula is used:
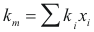
Where km is the thermal conductivity of the mixture (w/m°K)
ki is the thermal conductivity of the component i
xi is the fraction molar of the component i.
Latent heat of Vaporization
For a pure compound
to obtain the latent heat of vaporization the following formula is used:
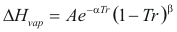
Where ΔHvap is the latent heat of vaporization (kj/mol)
A, α, β is constant that it depends on the compound
Tr is the reduced temperature
Enthalpy
For the liquids
to obtain the genitalia of a liquid the following formula is used:
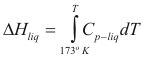 ole13.gif
ole13.gif
Where ΔHliq is the enthalpy of the liquid (cal/mol)
Cp-liq is the caloric capacity of the liquid (cal/mol°K)
T is the temperature of the liquid (°K)
For the gases
to obtain the enthalpy of a gas the following formula is used:
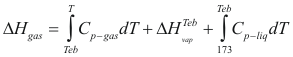
Where ΔHgas is the enthalpy of the gas (cal/mol)
Cp-gas is the caloric capacity of the gas (cal/mol°K)
ΔHvap is the latent heat of vaporization (kj/mol)
Cp-liq is the caloric capacity of the liquid (cal/mol°K)
T is the temperature of the liquid (°K)
Teb is the temperature of boil to a given pressure.
Factor ki - program online for pure compound
to obtain the factor ki of each compound is used their properties you criticize and value asentric:
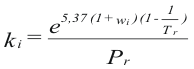
Where Pr is the reduced pressure
Tr is the reduced temperature
ω is the factor asentric
Factor Z - program online for pure compound
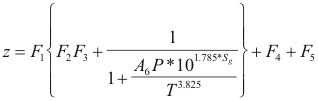
Where z is the factor of compressibility.
T is the temperature(°R)
P is the pressure (psi) (P <5000 psi)
Sg is the specifies gravity. (0.5 - 0.8)
F1 = P(0.251*Sg-0.15)-0.202*Sg+1.106
F2=1.4e-0.0054(T-460).
F3=A1*P5+A2*P4+A3*P3+A4*P2+A5*P
F4={0.154-0.152*Sg}*P(3.185*Sg-1)e-0.5*P-0.02
F5=0.35*{(0.6-Sg)E-1.039(P-1.8)2}
A1=0.001946
A2=-0.027635
A3=0.136315
A4=-0.23849
A5=0.105168
A6=3.44x108
Temperature of boil - program online for pure compound
to calculate the temperature of boil of a mixture the following equality should be completed:
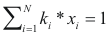
Where ki is the balance coefficient to a pressure and given temperature.
xi is the fraction molar of the compound i.
.
Condensation temperature - program online for pure compound
to calculate the temperature of condensation of a mixture the following equality should be completed:
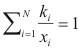
Where ki is the balance coefficient to a pressure and given temperature.
xi is the fraction molar of the compound i.
Chart for the conductivity thermal of the gas
| Formula | Compound | A | B | C | TMIN | TMAX |
| N2 | Nitrogen | 0.00283 | 8.125E-05 | -0.00000002 | ||
| CO2 | Dioxide Carbon | -0.012 | 0.00010208 | -0.000000022403 | 195 | 1500 |
| H2S | Sulfur hydrogen | |||||
| H2O | Water | 0.00193 | 4.52E-05 | 0.00000005 | ||
| C1 | Methane | -0.00935 | 0.00014028 | 0.00000003318 | 97 | 1400 |
| C2 | Ethane | -0.01936 | 0.00012547 | 0.00000003829 | 225 | 825 |
| C3 | Propane | -0.00869 | 6.6409E-05 | 0.00000007876 | 233 | 773 |
| i-C4 | i-Butane | -0.00115 | 1.4943E-05 | 0.00000014921 | 261 | 673 |
| n-C4 | n-Butane | -0.00182 | 1.9396E-05 | 0.00000013818 | 225 | 675 |
| i-C5 | i-Pentane | -0.00389 | 3.1816E-05 | 1.0396E-07 | 250 | 475 |
| n-C5 | n-Pentane | 0.00137 | 1.8081E-05 | 1.2136E-07 | 225 | 480 |
| C6 | n-Hexane | -0.002 | 7.779E-06 | 1.3824E-07 | 290 | 480 |
| C7 | n-Heptane | -0.00172 | 1.6565E-05 | 1.0525E-07 | 250 | 750 |
| C8 | n-Octane | -0.00213 | 1.8456E-05 | 9.4775E-08 | 300 | 800 |
| C9 | n-Nonane | -0.00655 | 3.2637E-05 | 7.715E-08 | 449 | 678 |
| C10 | n-Decane | -0.00113 | 8.109E-06 | 9.6092E-08 | 470 | 700 |
| C11 | n-Undecane | 0.01364 | -4.8303E-05 | 1.4396E-07 | 470 | 800 |
| C12 | n-Dozedecane | -0.00812 | 2.915E-05 | 7.2085E-08 | 489 | 1000 |
| C2H4O2 | Acid acetic | 0.00234 | -6.5956E-06 | 1.1569E-07 | 295 | 687 |
| C2H2 | Acetylene | -0.00358 | 6.2542E-05 | 7.0646E-08 | 200 | 600 |
| C3H6O | Acetone | -0.00084 | 8.7475E-06 | 1.0678E-07 | 273 | 572 |
| NH3 | Ammoniac | |||||
| CO | Monoxide Carbon | 0.00158 | 8.2511E-05 | -1.9081E-08 | 70 | 1250 |
| CH2CH2 | Ethylene | -0.00123 | 3.6219E-05 | 1.2459E-07 | 150 | 750 |
| O2 | Oxygen | 0.00181 | 8.575E-05 | -2E-08 | ||
| C4H8 | i-Butylene | -0.00327 | 3.0146E-05 | 1.2529E-07 | 250 | 850 |
| C3H6 | Propylene | -0.01116 | 7.5155E-05 | 6.5558E-08 | 250 | 1000 |
| CH3OH | Methanol | 0.00234 | 5.434E-06 | 1.3154E-07 | 273 | 684 |
| C2H5OH | Ethanol | -0.00556 | 4.362E-05 | 8.5033E-08 | 351 | 991 |
| C6H6 | Benzene | -0.00565 | 3.4493E-05 | 6.9298E-08 | 325 | 700 |
| neo-C5 | neo-Pentane | 0.00071 | -8.1028E-06 | 1.8759E-07 | 270 | 425 |
Chart for the conductivity thermal of the liquid
| Formula | Compound | A | B | C | TMIN | TMAX |
| N2 | Nitrogen | |||||
| CO2 | Dioxide Carbon | -1.3679 | 0.8092 | 304.19 | 217 | 289 |
| H2S | Sulfur hydrogen | |||||
| H2O | Water | -0.27070528 | 0.00456253 | -0.00000546 | ||
| C1 | Methane | -1.0976 | 0.5387 | 190.58 | 91 | 181 |
| C2 | Ethane | -1.3474 | 0.7003 | 305.42 | 90 | 290 |
| C3 | Propane | -1.2127 | 0.6611 | 369.82 | 85 | 351 |
| i-C4 | i-Butane | -1.6862 | 0.9802 | 408.14 | 114 | 388 |
| n-C4 | n-Butane | -1.8929 | 1.2885 | 425.18 | 135 | 404 |
| i-C5 | i-Pentane | -1.6824 | 0.9955 | 460.43 | 113 | 437 |
| n-C5 | n-Pentane | -1.2287 | 0.5322 | 469.65 | 143 | 446 |
| C6 | n-Hexane | -1.8389 | 1.186 | 507.43 | 178 | 482 |
| C7 | n-Heptane | -1.8482 | 1.1843 | 540.26 | 183 | 513 |
| C8 | n-Octane | -1.8388 | 1.1699 | 568.83 | 216 | 540 |
| C9 | n-Nonane | -1.7865 | 1.1033 | 595.65 | 220 | 566 |
| C10 | n-Decane | -1.7768 | 1.0839 | 618.45 | 243 | 588 |
| C11 | n-Undecane | -1.6318 | 0.9325 | 638.76 | 248 | 607 |
| C12 | n-Dozedecane | -1.7989 | 1.1109 | 658.2 | 264 | 625 |
| C2H4O2 | Acid acetic | -1.2836 | 0.5893 | 592.71 | 290 | 563 |
| C2H2 | Acetylene | |||||
| C3H6O | Acetone | -1.3857 | 0.7643 | 508.2 | 178 | 483 |
| NH3 | Ammoniac | |||||
| CO | Monoxide Carbon | -1.7115 | 1.1359 | 132.92 | 68 | 126 |
| CH2CH2 | Ethylene | -1.3314 | 0.8527 | 282.26 | 104 | 268 |
| O2 | Oxygen | |||||
| C4H8 | i-Butylene | -1.49020 | 0.8491 | 417.90 | 133 | 397 |
| C3H6 | Propylene | -1.43760 | 0.7718 | 364.76 | 88 | 347 |
| CH3OH | Methanol | -1.1793 | 0.6191 | 512.58 | 175 | 487 |
| C2H5OH | Ethanol | -1.3172 | 0.6987 | 516.25 | 159 | 490 |
| C6H6 | Benzene | -1.6846 | 1.052 | 562.16 | 279 | 534 |
| neo-C5 | neo-Pentane | -1.7534 | 1.0306 | 433.78 | 257 | 412 |
BACK